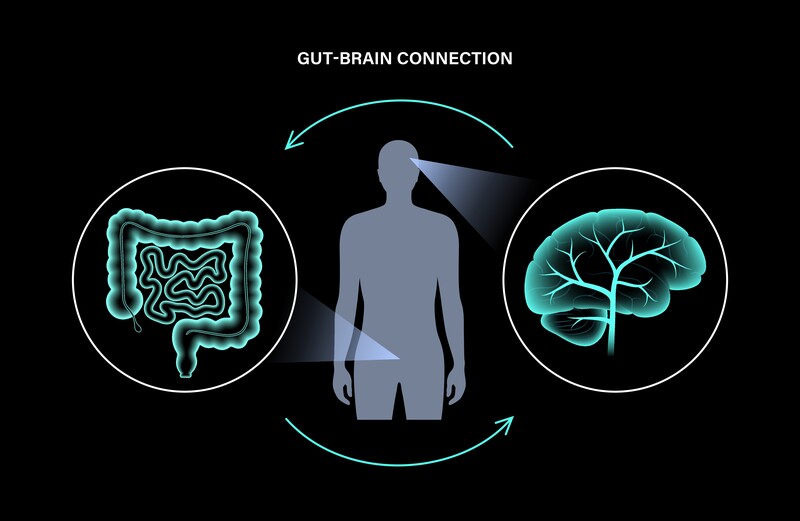
Link between Alzheimer’s disease and gut microbiota
Numerous medical studies have supported the idea that gut inflammation can lead to problems of mood and memory as well as other neurologic symptoms including Alzheimer’s Disease and dementia. In many of our articles, we have shown recent studies that a healthy gut microbiota (the collective name for the bacteria that live in the digestive/intestinal tract) can protect many people from developing certain diseases including cancer and various bowel related syndromes. The idea that probiotics, the “good” bacteria, promotes a healthy gut microbiota and supports our cognitive system by regulating inflammation is a widely held scientific belief.
Folate, Vitamin B12 and the Gut Microbiome
How does gut inflammation impact memory cognitive function? It is a very complex issue. Sometimes understanding how the gut works can offer us clues as to how it does not work. In one study the gut does not work well is when it is deficient in Folate and vitamin B12. When the gut is deficient in these nutrients, memory becomes a problem.
In December 2019, a team of university researchers published the results of their study (1) on how folate (folic acid is one form of folate) and vitamin B12 deficiencies worsened memory function and disturbed the gut microbiota in amyloid-β infused (Alzheimer’s induced) rats.
The learning points of this research were:
Folate and vitamin B12 deficiencies are associated with metabolic diseases that may impair memory function.
Folate and vitamin B12 deficiencies and a high-fat diet exacerbated memory impairment. (The laboratory rats could not or had difficulty remembering how to get out of a water maze).
Folate and vitamin B12 may differently alter mild cognitive impairment, glucose metabolism, and inflammation by modulating the gut microbiome in rats with Alzheimer’s disease (AD)-like dementia.
The key to this research was that there was an alteration in the way the gut works and this led to a problem in the brain. Specifically the development of Alzheimer’s disease.
Gut dysbiosis (a reduction in the gut microbes)
Building on research like that above, doctors published research in the February 2021 issue of the journal Nutrients: (2)
The gut microbiota (GM) represents a diverse and dynamic population of micro-organisms and about 100 trillion symbiotic microbial cells that dwell in the gastrointestinal tract. Studies suggest that the gut microbiota can influence the health of the host, and several factors can modify the gut microbiota composition, such as diet, drug intake, lifestyle, and geographical locations.
Gut dysbiosis (a reduction in the gut microbes) can affect brain immune homeostasis through the microbiota-gut-brain axis and can play a key role in the pathogenesis of neurodegenerative diseases, including dementia and Alzheimer’s disease. The relationship between gut dysbiosis and Alzheimer’s disease is still elusive, but emerging evidence suggests that it can enhance the secretion of lipopolysaccharides (simply in this context a pro-inflammatory) and amyloids (abnormal proteins) that may disturb intestinal permeability and the blood-brain barrier. In addition, it can promote the hallmarks of Alzheimer’s disease, such as oxidative stress, neuroinflammation, amyloid-beta formation, insulin resistance, and ultimately the causation of neural death. Poor dietary habits and aging, along with inflammatory responses due to dysbiosis, may contribute to the pathogenesis of Alzheimer’s disease. Thus, gut microbiota modulation through diet, probiotics, or fecal microbiota transplantation could represent potential therapeutics in Alzheimer’s disease.
The “Lost-Link” to preventing Alzheimer’s disease?
A December 2019 study in the Journal of Alzheimer’s disease (3) suggested that gut inflammation and the regulation of brain-gut-microbiome axis may be a “lost link” in understanding and treating Alzheimer’s disease. The researchers noted:
“Despite extensive research into Alzheimer’s disease pathogenesis and therapeutic targets, there remains no breakthroughs in its management. In recent years, there has been a spark of interest in the connection between the brain and gastrointestinal tract, referred to as the brain-gut axis, and its potential implications for both metabolic and neurologic disease. Moreover, the gastrointestinal flora, referred to as the microbiome, appears to exert significant influence over the brain-gut axis.
With the need for expanded horizons in understanding and treating Alzheimer’s disease, many have turned to the brain-gut-microbiome axis for answers. . . we conclude with a brief description of documented clinical support for brain-gut-microbiome axis alteration in Alzheimer’s disease, including potential microbiome-based therapeutics for Alzheimer’s disease. Collectively, these findings suggest that the brain-gut-microbiome axis may be a “lost link” in understanding and treating Alzheimer’s disease and call for future work.”
The key findings of this research is, if you treat the gut, and you create harmony and balance in the gut, you may be treating Alzheimer’s disease.
A March 2019 study in the journal Molecular neurobiology (4) takes this one step further.
Alzheimer’s disease may begin in the gut
“One of the most important scientific discoveries of recent years was the disclosure that the intestinal microflora takes part in bidirectional communication between the gut and the brain. Scientists suggest that human gut microflora may even act as the “second brain” and be responsible for neurodegenerative disorders like Alzheimer’s disease.
Brain destructive mechanisms, that can lead to dementia and Alzheimer’s disease, start with the intestinal microbiome dysbiosis, development of local and systemic inflammation, and dysregulation of the gut-brain axis. . . It is strongly postulated that Alzheimer’s disease may begin in the gut, and is closely related to the imbalance of gut microbiota. This is promising area for therapeutic intervention. Modulation of gut microbiota through personalized diet or beneficial microbiota intervention, alter microbial partners and their products including amyloid protein, will probably become a new treatment for Alzheimer’s disease.”
Do the Bugs in Your Gut Eat Your Memories?
Do the Bugs in Your Gut Eat Your Memories? That was the title of a November 2020 review study published in the journal Brain sciences.(4) In this research the authors wrote:
“The gut microbiota is composed of dynamic communities of microorganisms that communicate bidirectionally with the brain via cytokines (cells that take part in the inflammatory response), neurotransmitters, hormones, and secondary metabolites, known as the gut microbiota-brain axis.The gut microbiota-brain axis is suspected to be involved in the development of neurological diseases, including Alzheimer’s disease, Parkinson’s disease, and Autism Spectrum Disorder. Alzheimer’s disease is an irreversible, neurodegenerative disease of the central nervous system, characterized by amyloid-β plaques, neurofibrillary tangles, and neuroinflammation.”
What we want to keyword in on is “neuroinflammation.” Let’s continue:
“Microglia (a type of neuroglia or glial cell that make up 10–15% of all cells found in the brain, act as the main form of active immune defense protecting the neurons of the brain and the spinal cord) and astrocytes (a cell that has many functions but here we focus on protecting the brain and the blood-brain barrier immune defense), the resident immune cells of the central nervous system, play an integral role in Alzheimer’s disease development, as neuroinflammation is a driving factor of disease severity. The gut microbiota-brain axis is a novel target for Alzheimer’s disease therapeutics to modulate critical neuroimmune and metabolic pathways. Potential therapeutics include probiotics, prebiotics, fecal microbiota transplantation, and dietary intervention.”
The suggest is then to target neuroimmune and metabolic pathways of the gut.
The inflammation and diet theory of Alzheimer’s Disease
A September 2019 paper (6) Alzheimer’s disease wrote: “Due to the fact that there is still no cure for Alzheimer’s dementia and available treatment strategies bring only symptomatic benefits, there is a pressing demand for other effective strategies such as diet. Since the inflammation hypothesis gained considerable significance in the Alzheimer’s disease pathogenesis, elucidating the modulatory role of dietary factors on inflammation may help to prevent, delay the onset and slow the progression of Alzheimer’s disease. Current evidence clearly shows that synergistic action of combined supplementation and complex dietary patterns provides stronger benefits than any single component considered separately. Recent studies reveal the growing importance of novel factors such as dietary advanced glycation end products (d-AGE), gut microbiota, butyrate and vitamin D3 on inflammatory processes in Alzheimer’s dementia.”
- Dietary advanced glycation end products (d-AGE) are harmful food based compounds. Your body is always trying to get rid of them. Also known as Advanced glycation end products, these compounds form when a protein or a fat combines with a sugar. The binding process is called glycation. Foods cooked at high temperature are typically high in these compounds. People with diabetes or high blood sugars are at a higher risk of producing too many advanced glycation end products which can accumulate in the body.
Exploring your options
At the Magaziner Center for Wellness, we approach disease and imbalance by first determining all contributing factors and creating an individualized, patient-centered treatment plan. The latest research has shown that there are a host of factors that can cause and contribute to Alzheimer’s- from environmental toxins to nutrition to heavy metal toxicity and more. Every person has a unique biochemistry which reacts to pollutants and toxins differently, and requires an individualized course of treatment. While the disease may look similar from person to person, we are not treating the disease. We are healing the individual.
In order to determine the unique contributing factors of each individual, we utilize extremely thorough blood and urine tests, as well as a complete examination of every aspect of the body, from mitochondrial function to heavy metal toxicity and more. We then create a personalized care plan based on these findings which may include treatments such as chelation therapy, hyperbaric oxygen therapy, antioxidant nutritional supplements, intravenous vitamins, diet and nutrition.
If you would like to explore more information, please contact our office so we can start a conversation with you.
Related articles:
Does Caloric Restriction Prevent Memory Loss?
Research on Vitamin B12 and Folic Acid Deficiency Connected to Alzheimer’s Disease and Dementia
Post-Menopausal Memory Loss
Green Tea and Alzheimer’s Disease
Preventing Vascular Dementia
Iron Toxicity: A Cause of Alzheimer’s Disease
References
1 Park S, Kang S, Kim DS. Folate and vitamin B-12 deficiencies additively impaired memory function and disturbed the gut microbiota in amyloid-β infused rats. International Journal for Vitamin and Nutrition Research. 2019 Dec 16.1: Sun M, Ma K, Wen J, Wang G, Zhang C, Li Q, Bao X, Wang H.
2 Shabbir U, Arshad MS, Sameen A, Oh DH. Crosstalk between gut and brain in Alzheimer’s disease: the role of gut microbiota modulation strategies. Nutrients. 2021 Feb;13(2):690.
3 Sun M, Ma K, Wen J, Wang G, Zhang C, Li Q, Bao X, Wang H. A review of the brain-gut-microbiome axis and the potential role of microbiota in Alzheimer’s disease. Journal of Alzheimer’s Disease. 2020 Jan 1(Preprint):1-7.
4 Sochocka M, Donskow-Łysoniewska K, Diniz BS, Kurpas D, Brzozowska E, Leszek J. The Gut Microbiome Alterations and Inflammation-Driven Pathogenesis of Alzheimer’s Disease-a Critical Review. Mol Neurobiol. 2019 Mar;56(3):1841-1851. doi: 10.1007/s12035-018-1188-4. Epub 2018 Jun 23. PMID: 29936690; PMCID: PMC6394610. -1232
5 Borsom EM, Lee K, Cope EK. Do the Bugs in Your Gut Eat Your Memories? Relationship between Gut Microbiota and Alzheimer’s Disease. Brain sciences. 2020 Nov;10(11):814.
6 Szczechowiak K, Diniz BS, Leszek J. Diet and Alzheimer’s dementia–Nutritional approach to modulate inflammation. Pharmacology Biochemistry and Behavior. 2019 Sep 1;184:172743.































Recent Comments